 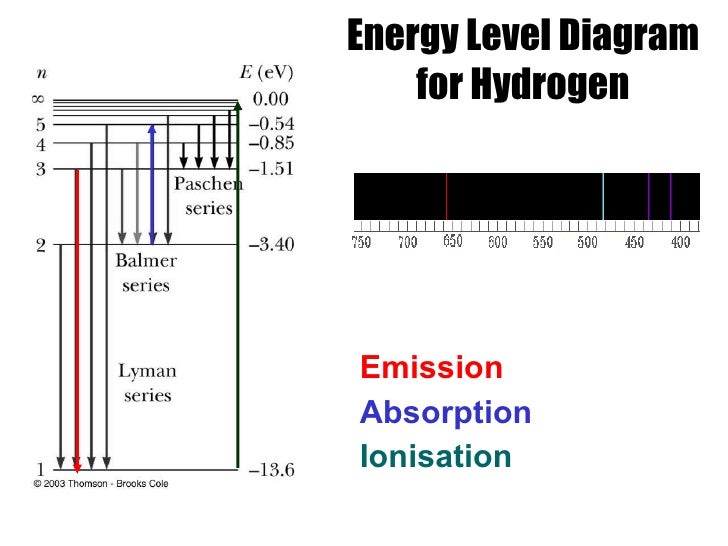
Universality of Physical Laws in Space and Time You can also see how the number of atoms in a given state Use the UNL Astronomy Education program's Hydrogenįurther explore how emission and absorption lines are created (link will appear Rainbow of colors of a continuous spectrum. High enough to where the smeared lines all merge together to produce the Of smearing, or broadening, depends on the density. Orange line is fattened so that one edge is in the yellow wavelengths and the This smears out the normally sharp spectral lines (they 
The energy levels of the atoms are distorted by their neighboringĪtom's electrons. Photons with energies between 1.89 and 3.4 eV not listed above will NOT be absorbed by the atom.Ī thermal spectrum is produced by atoms that are closely packed 
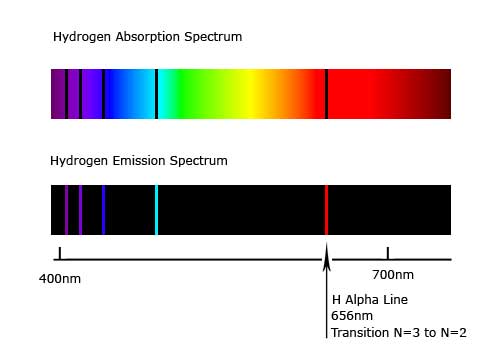
The line at 2.86 eV is for the n=2 to n=5 hop and the line at 3.02 eV is for the n=2 to n=6 hop. The spectrum shows the hydrogen absorption line energies.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
